- ACTIVE INGREDIENT NSC 631570 :
ONLY KILLS CANCER CELLS - HEALTHY CELLS STAY HEALTHY - I. EFFICACY
- II. SAFETY
- III. QUALITY
- BIBLIOGRAPHY
NSC 631570 : EFFICACY, SAFETY, QUALITY
INTRODUCTION: THE FIRST MEDICAMENT THAT KILLS CANCER CELLS BUT NOT HEALTHY CELLS
Cancer cells can be killed without damaging healthy cells
A new preparation was presented at the 13th International Congress of Chemotherapy in Vienna in August-September 1983 – thiophosphoric acid derivative of alkaloids from greater celandine (NSC631570). The development of this preparation was the first significant step on the way to solving the problem. In vitro tests showed that after incubation with NSC631570 normal liver cells and Ehrilch tumour ascitic cells demonstrate different oxygen consumption: after initial increase, oxygen consumption in cancer cells falls to zero whereas oxygen consumption in normal cells returns to normal and the cells remain undamaged (38). This study brought the first indications that, in contrast to its starting substances thiotepa (a well known cytostatic agent) and greater celandine alkaloids, NSC631570 is in fact only toxic against cancer cells and not against normal cells.
The second indication was provided by clinical use, where NSC631570 caused no noteworthy side-effects (21). It improved patients’ general condition as well as their immune status which had previously been impaired by chemotherapy (22).
The third indication was provided by a study at the University of Miami, based upon which the therapeutic index of NSC631570 was calculated to be 1250 (26). This is unusually high for an anti-cancer preparation. The therapeutic index is the ratio of the toxic dose to the therapeutic dose and reflects the level of safety of a medicine. The therapeutic index of conventional cytostatic preparations is in the range of 1. 4-1.8 meaning that an overdose can have fatal consequences. There is no risk of an overdosage with NSC631570 on account of its very high therapeutic index of 1250.
The development of NSC631570 was a trail-blazing discovery. The preparation has shown that the problem can be solved and has changed our ideas about healthy and cancer cells.
The presentation at the congress was greeted both by scepticism and great interest. Many reputed research institutes such as the National Cancer Institute (USA), EORTC, the University of Miami, the Rochester University (USA), and University of Tübingen (Germany) began to test the preparation to better explain its unique properties and anticancer potential. In the NCI test model, in contrast to conventional cytostatic preparations which caused the growth inhibition only in some cancer cell lines, like thiotepa inhibited the growth of MLI-09 (non-small lung cancer) and UOK-57LN (renal cancer), NSC631570 killed all 60 tested cancer cell lines (190), which represent the eight important human tumours, including the cell lines which were resistant to the strongest cytostatic drug at the time, cisplatin.
This generated still more interest in scientific circles. Leading scientists examined NSC631570, each group with the method available. Thanks to this variety of experimentation the fine mechanisms of action of NSC631570 at different levels could be deciphered: firstly at cellular level with oxygen consumption (effect on mitochondria; 38), then on the level of chromosomes (effect on DNS and RNS; 58, 63), cell organelles and molecules (43, 62). These experiments produced extremely interesting results and not only confirmed the selective effect of NSC631570 but thoroughly destroyed all doubt on the matter. This means that NSC631570 can differentiate between healthy and malignant cells, a feature not yet managed by other anti-cancer drugs. Interest in NSC631570 is growing and research is continuing (261-266).
Researchers at the University of Natural Resources and Life Sciences, Vienna compared NSC631570’s inhibitive effect on the reproduction of malignant and normal cells. In order to achieve 50% growth inhibition a tenfold concentration of NSC 631570 had to be administered to normal endothelial cells in comparison to a human osteosarcoma cell line. Laser scanning microscopy showed a high capacity to absorb NSC631570 in malignant cells while absorption in normal cells was considerable lower under the same experimental conditions (36).
In a study of the effect of NSC631570 on K-562 erythroleucemia cells it was found that the preparation causes bimodal cell death. At lower NSC631570 concentrations malignant cells die as a result of apoptosis, at higher concentrations the formation of microtubules is prevented und polyploidy occurs (62).
In 1998 a group around Anne Panzer (University of Pretoria, South Africa) proved the selective effect of NSC631570 on molecular level. Tests on human cervical carcinoma cells HeLa, squamous cell carcinoma WHCO5 and normal equine lung cell lines demonstrated that NSC631570 is selectively toxic against cancer cells. It causes a metaphase block which is characterised by an abnormal distribution of chromosomes and the formation of micronuclei and results in apoptosis. Normal cells are not influenced in the process (139).
In 2000 in a study of cell proliferation after absorption of BrdU in the cell lines AsPC1, BxPC3, MiaPaCa2, Jurkat and THP-1 and the cell cycle phases – with the help of Giemsa staining, researchers from Ulm found that 10 µg/ml NSC631570 causes a clear accumulation of cancer cells in phase G2/M. Interestingly no difference was seen in the rate of apoptosis in normal peripheral mononuclear cells treated with NSC631570 compared to those untreated. The blastogenic response of mitogen-stimulated lymphocytes was even significantly increased. The authors showed that NSC631570 blocks pancreas cancer cells in the prophase by inhibiting tubulin polymerisation (181). This study confirmed that NSC631570 exerts no influence on normal cells.
Also in 2000 researchers at Rochester University (USA) examined the effect of NSC 631570 on concentrations of cyclins and cyclin-dependent kinases in epidermoid carcinoma cell lines ME180 and A431 as well as the prostate cancer cell line LNCaP. Changes were found in the concentrations of mitotic cyclins A and B1 as well as CDK1 and CDK2. The researchers also observed increased expression of the CDK-inhibitor p27 in both cell lines, which led to the accumulation of cancer cells but not normal cells in the G2/M phase (147, 149).
In another study from 2000 entitled „NSC631570TM, a semisynthetic Chelidonium majus alkaloid derivative, acts by inhibition of tubulin polymerization in normal and malignant cell lines“ (Cancer Letters 160 (2000) 149-157) researchers from South Africa found that NSC631570 inhibits the polymerisation of tubulin (185). The authors used human fibroblasts as a normal cell line.
Normal human fibroblasts were later used in the tests by the scientists of the Eberhard-Karls-University Tuebingen (Germany) as well as of the Instituto Nacional de Cancerologia (Mexiko City, Mexiko), also. Both research teams could not find any toxic effects of NSC631570 on these normal cell lines. Moreover, A. Panzer et al noted in their article: „Both NSC631570TM and chelidonine had weak activity in this system…“ (185).
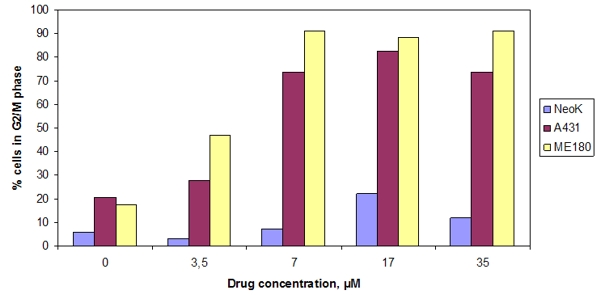
Fig. 1. Effect of different NSC631570 concentrations on G2/M cell population. Cells were treated with the indicated concentrations for 24 h and analyzed for DNA content by flow cytometry. The data presented are the percentage of NeoK (normal human keratinocytes), A431 and ME180 (epidermoid carcinoma cells) cell populations in G2/M phase. From Roublevskaia et al, 2000 (147).
In 2002 scientists at the Eberhard-Karls-University Tuebingen (Germany) examined the effect of NSC 631570, alone or combined with radiation (1-10 Gy), on cell survival, the modification of the cell cycle and the induction of apoptosis in the exponentially growing human tumour cell lines MDA-MB-231 (breast cancer), PA-TU-8902 (pancreatic cancer), CCL-221 (colonic cancer), U-138MG (glioblastoma) and the human skin fibroblasts HSF1, HSF2 and lung fibroblasts CCD32-LU. Without radiation NSC 631570 had a time and dose dependent cytotoxic effect which was more pronounced in cancer cells than in normal cells. Combined with radiation NSC 631570 resulted in increased cytotoxicity against colonic cancer and glioblastoma cell lines but not against breast cancer and pancreatic cancer cell lines. By means of flow cytometry it was shown that NSC 631570 modulated the toxic effect of radiation on these human cancer cell lines by causing their accumulation in the G2/M phase of the cell cycle. Its protective effect on normal human fibroblasts speaks in favour of its use in combined radio-chemotherapy (184).
In 2005 the ability of NSC631570 to induce apoptosis was studied at the University Hospital Tuebingen (Germany) on a Jurkat lymphoma model. NSC631570 proved itself to be a strong inducer of apoptosis. More detailed investigations showed that it caused the depolarisation of mitochondrial membranes and as a result the activation of caspases (246).
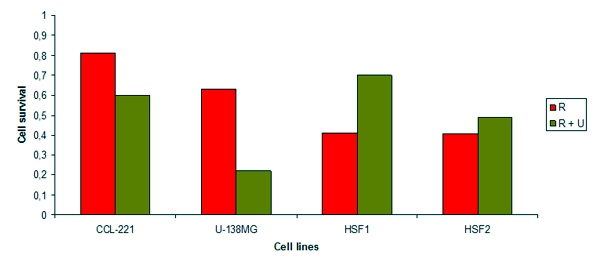
Fig. 2. Survival of irradiated cells with and without NSC631570 treatment. R – irradiation, 2 Gy; R + U – irradiation, 2 Gy and treatment with NSC631570. Cell lines: CCL-221 - colorectal carcinoma, U-138MG - glioblastoma, HSF1 und HSF2 - normal fibroblasts. After Cordes et al, 2002 (184).
In 2006 researchers at the Instituto Nacional de Cancerologia (Mexico City, Mexico) found that NSC631570 triggers apoptosis in a series of cancer cell lines (human cervical carcinoma HeLa, HeKB, HeKS32, HeBcll3, HeNFR and HelKK, colonic cancer SW480, kidney cancer HEK293, osteosarcoma MG 63) by activating the intrinsic cell death pathway. Interestingly the non-transformed fibroblast cell line hTERT was not sensitive to this drug (255).
At the University of Pisa (Italy) the cytotoxic effect of NSC 631570 was examined on two primary pancreatic cancer cell lines (PPTCC), fibroblasts from ductal pancreatic cancer tissue samples (F-PDAC) and an immortalised ductal epithelial pancreas cell line (HPNE). Cytotoxicity was established by means of CellTiter 96 kit. The modulation of NSC 631570 absorption in the medium was determined with the help of the fluorescence of NSC 631570 under UV light. The cytotoxic effect of NSC 631570 on pancreatic cancer cell lines was considerably higher than on the fibroblasts and epithelial cells (20% as against 80% living cells). In addition the fluorescence test showed that PPTCC cells absorbed more of the preparation than F-PDAC and HPNE cells. These results show a selective effect of NSC 631570 on the pancreatic cancer cells, which indicates different transport systems or a higher rate of metabolism of the preparation in the PDAC cells (265).
As can be seen, many aspects of the effect of NSC631570 on cancer and normal cells have so far been studied. Nevertheless, the possibility exists that these effects are merely the results of an as yet unknown process which NSC631570 induces in cancer cells but not in normal cells. As NSC631570 killed in the tests almost all cancer cell lines, it can be suggested that a factor occurs when a normal cell becomes a cancer cell. This factor is affected with NSC631570. If this phenomenon can be deciphered it could provide very important indicators for the significant difference between normal cells and cancer cells, point towards the real cause of cancer and open up completely new perspectives for the development of new anti-cancer drugs, not only for treatment but also for use in cancer prevention.
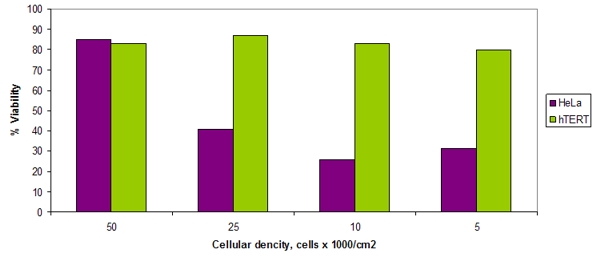
Fig. 3. Effects of NSC631570 on cervical carcinoma cell line HeLa and on the normal fibroblasts hTERT expressed as percentage of living cells after 48 h incubation with NSC631570 40 µg/ml. From Mendoza et al, 2006 (255).
These studies on the selective effect of NSC631570 clearly revealed cancer cells can be killed without any damage to the health tissues.
Affinity of NSC 631570 to the cancer cells
It is well known all living cells have a membrane potential of about -60 to –100mV. The negative sign of the membrane potential indicates that the inside surface of the cell membrane is relatively more negative than the than the immediate exterior surface of the cell membrane.
As far back as 1938 Dr. Paul Gerhardt Seeger originated the idea that destruction or inactivation of enzymes, like cytochrome oxidase, in the respiratory chain of the mitochondria was involved in the development of cancer.
1948 G.R. Mider and co-workers revealed the cancer cells are more negatively charged than normal cells.
During the production of NSC631570 celandine alkaloids become positively charged. This explains the high affinity of NSC631570 to the cancer cells.
Researchers at the University of Natural Resources and Life Sciences, Vienna (Austria) revealed human osteosarcoma cells to have a high capacity to absorb NSC631570 while absorption in normal endothelial cells was considerable lower under the same experimental conditions (36).
This selective uptake of NSC631570 in the cancer cells has been confirmed due to its unique property to autofluorescence under UV light (5).
A study examined how NSC 631570 affects the electrokinetic potential (EKP) of malignant and benign cells. The EKP of the Ehrlich’s carcinoma cells dropped after incubation with NSC 631570. The EKP decrease of normal cells was less pronounced (221).
The cytotoxic effect of NSC 631570 was examined on two primary pancreatic cancer cell lines (PPTCC), fibroblasts from ductal pancreatic cancer tissue samples (F-PDAC) and an immortalised ductal epithelial pancreas cell line (HPNE). Cytotoxicity was established by means of CellTiter 96 kit.
In the tests with primary pancreatic cancer cell lines the modulation of NSC 631570 absorption in the medium was determined with the help of the fluorescence of NSC 631570 under UV light. The fluorescence test showed that carcinoma cells absorbed more of the preparation than fibroblasts and normal epithelial cells (265).
The selective accumulation of NSC 631570 in the cancer cells explains its favourable safety profile and high therapeutic index of 1250.
Unusual for an anticancer agent NSC 631570 possesses some distinct immune properties (24, 44). It was Prof. Andrejs Liepins of the St. John’s Memorial University, St. John’s, Canada who first pointed to this interesting fact. In the work with the C57BL/6 mice he revealed NSC631570 to be an effective biological response modifier (BRM). After incubation with NSC631570 the lytic activity of the splenic lymphocytes from the alloimmunised mice increased up to 48 fold (fig. 4). On the day 18 this increase was the highest and then faded slightly (fig. 5). The lytic activity of the interleukin-2 treated spleen cells and of the peritoneal exudate lymphocytes increased as well (17).
In several immune target-effector systems NSC 631570 significantly amplified the malignotoxic activity of macrophages (231), lymphocytes and NK cells (47), and stimulates dendritic cells maturation in vitro (258). While the parameters like B-lymphocytes count, immune globulin concentrations, complement and acute phase proteins did not changed significantly, it can be postulated NSC 631570 modulates the cellular part of the immune system whereas the humoral part remains unaffected.
Human lymphocytes as well as guinea pig lymphocytes were activated more pronounced when incubated with NSC631570 than with phytohemagglutinin. In rats NSC 631570 caused a clear increase of macrophages with NK-activity (49).
It was revealed in the tests on CBA mice and Wistar rats that NSC 631570 stimulates macrophages. As marker of this activity the enzyme chitotriosidase, a part of the native immunity was used (168).
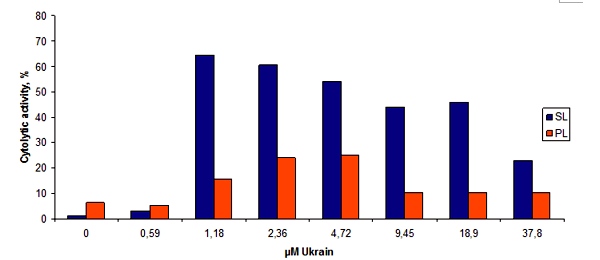
Fig. 4. The effect of NSC631570 on the cytolytic activity of spleen (SL) and peritoneal (PL) lymphocytes from alloimmunized mice. From Liepins et al, 1992 (17).
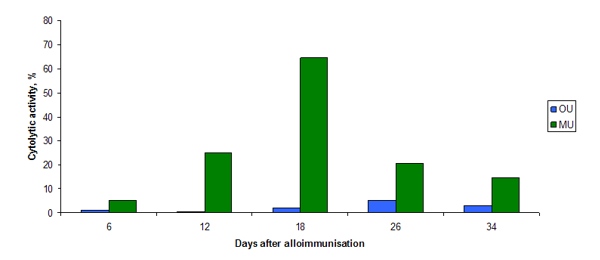
Fig. 5. The effect of 1.18 µM NSC631570 on the cytolytic activity of spleen cells harvested at various time intervals after autoimmunization. From Liepins et al, 1992 (17).
In a study on intact and thymus-ectomised mice NSC 631570 augmented the endocrine function of thymus. After NSC 631570 administration, increased production of substances with thymosin-like activity was detected. Repeated administration of NSC 631570 caused a 2fold rise of T-cells in blood, a 4.5fold rise of large granulocytes and increase of the NK-activity of splenocytes. The production of interferon and antibodies after the antigen administration was increased as well (180).
The effect of well-known immune modulators interferon-gamma, NSC 631570 and pokeweed mitogen on the selective uptake of technetium-99m(99mTc)-labelled tumor necrosis factor (TNF) was studied in the intramuscular implanted murine embryonic carcinoma. The highest absolute tumor uptake of 99mTc- TNF was achieved when NSC 631570 was used, followed by IFN-γ and pokeweed mitogen (fig. 6; 104).
In the experiments on BALB/c and F1 (BALB/c x C57BL/6J) mice it was revealed that NSC 631570 inhibits the allergic sensitization of animals against ovalbumin, expressed in the weakened IgE-reaction and decreased histamine release. The incubation of ovalbumin with NSC 631570 induced decreased antigenicity of this protein (84).
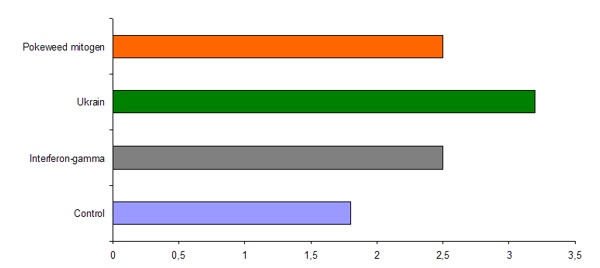
Fig. 6. Absolute (% administered dose/g) tumor uptake of -TNF-α in mice BALB/c 1,5 h after administration. After Thakur et al, 1992 (104).
The immune modulating effect of NSC 631570 was studied in several studies in mice. Repeated subcutaneous injections of NSC 631570 to mice infected with the twofold LD50 of E. coli, S. aureus, or influenza virus increased the survival rate of the animals significantly (60, 87, 89).
When human lymphocytes were incubated with phytohemagglutinin (PHA) and NSC 631570, increased absorption of 3H-thymidin in the cells was observed. The authors point out the strong synergetic effect of NSC 631570 and phytohemagglutinin (76).
By means of the cell proliferation assay the mitogenic effects of PHA and NSC 631570 on human peripheral blood mononuclear cells (PBMC) were studied. It was revealed even a short pretreatment of the PBMC with NSC 631570 has a strong synergetic effect on the PHA-mitogenesis. Consequently, the cell stimulation parameters were much higher after combined stimulation than after using PHA alone (65).
In experiments with murine (CC57 Black/6) macrophages and rabbit G-actin, the effects of NSC 631570 and sanguinarine on phagosome-lysosome membrane fusion and actin cytoskeleton were studied. The most stimulating effect on the phagosome-lysosome fusion exerted sanguinarine at 10 µmol and NSC 631570 at 5 µmol. At the same dose NSC 631570 doubled the content of fibrillary actin in murine peritoneal macrophages. Moreover, NSC 631570 and sanguinarine induced the polymerisation of rabbit globular actin. These effects were dose-dependent. The authors suggest sanguinarine and NSC 631570 can alter intracellular membrane transport (231).
The publications mentioned here point out that NSC631570 due to its many beneficial immune properties can be categorized as one of the best immune modifier.
Another important feature of NSC631570 is the inhibition of the formation of the new blood vessels supplying a tumor. Due to these antiangiogenic properties NSC631570 administered before surgery brings about better demarcation of the tumor from surrounding tissue and the tumor encapsulation. This alleviates the surgical removal of tumors what has been confirmed in breast cancer studies (68-73, 114). In tests in vitro, NSC631570 inhibited in a dose-dependent manner the proliferation of human endothelial cells without exerting cytotoxic effect. The angiogenesis inhibition was observed on the capillary formation model (136). This inhibition of the neoangiogenesis prevents the metastasis formation as well.
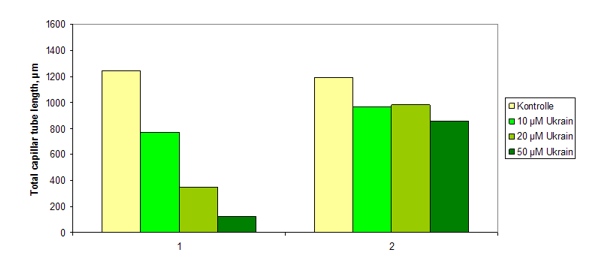
Fig. 7. The length of the total capillary tubes after the incubation with NSC631570 in given concentrations. 1 – HUVEC were incubated with NSC631570 for 4 h. 2 – HUVEC were preincubated with NSC631570 for 4h and then incubated for 2 h in fresh medium without NSC631570. HUVEC – human umbilical vein endothelial cells. After Koshelnick et al, 1998 (136).
